
Despite early postoperative worsening of DC function, long-term follow-up showed significant recovery and improvement in quality of life. After myelotomy, 3 patients experienced > 50% reduction in amplitude of SSEPs. In 92% of cases, the AM and neurophysiologic midlines were concordant. SCS was less time-consuming ( p = 0.001), cheaper, and easier to handle. If baseline SSEPs were poor, both methods were limited. If both recordings could be acquired (6/13), concordance was 100%. SCS was applicable in all patients with determination of the midline in 9/13. The DCM electrode detected the midline in 9/13 patients with handling limitations in the remaining patients. Standardized neurological examinations were performed preoperatively, 1 week postoperatively and after more than 12 months. Procedural and technical aspects were compared. SSEPs at the DC after stimulation of tibial/median nerve with an 8-channel DCM electrode and cortical SSEP phase reversal at C3/C4 after SCS using a bipolar concentric probe were recorded. Patients with surgically treated imSCT were prospectively included between 04/2017 and 06/2019. We compare application and feasibility of both methods. Dorsal column mapping (DCM) and spinal cord stimulation (SCS) can identify DC neurophysiologically.
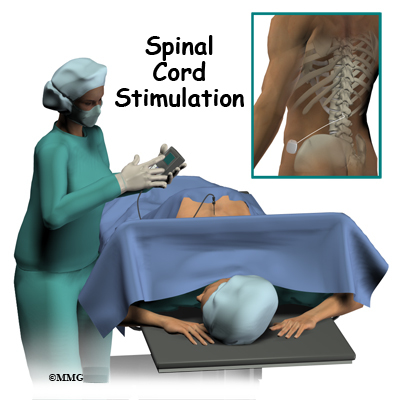
The SCS system when programmed to a frequency of 10 kHz, may help treat chronic, intractable pain in a patient’s low back, trunk or limbs, when there is no prior back surgery and the patient is not a candidate for back surgery.In surgery for intramedullary spinal cord tumors (imSCT), distortion of the anatomy challenges the visual identification of dorsal columns (DC) for midline myelotomy. The SCS system when programmed to a frequency of 10 kHz, is used as an aid to manage chronic pain in the trunk or limbs, including one-sided or two-sided pain associated with failed back surgery syndrome, intractable low back pain, leg pain, pain associated with nerve damage caused by diabetes ( diabetic neuropathy), and refractory back pain when the patient is not a candidate for back surgery and back surgery hasn’t been performed previously. The external remote control is battery operated and can be controlled by the patient or a health care provider. The signals tell the signal generator when to deliver appropriate stimulation to the spinal cord. The implanted signal generator receives radio signals from the remote control. This supplement expands the Senza SCS systems’ Indications for Use when programmed to a frequency of 10 kHz, to aid in the management of intractable back pain when the patient is not a candidate for back surgery, and back surgery hasn’t been performed previously. The main components of the SCS system include an implanted signal generator that is connected to one or two implanted leads and a remote control that can turn the stimulator ON or OFF to allow adjustments of therapy settings. The Senza Spinal Cord Stimulation (SCS) system is an implanted, rechargeable spinal cord stimulation system intended to treat long-term (chronic) pain in the trunk or limbs that is difficult to manage (intractable). Product Name: Senza Spinal Cord Stimulation (SCS) SystemĪddress: 1800 Bridge Parkway Redwood City, CA 94065Īpproval Letter: Approval order What is it? See the links below to the Summary of Safety and Effectiveness Data (SSED) and product labeling for more complete information on this product, its indications for use, and the basis for FDA’s approval. This is a brief overview of information related to FDA’s approval to market this product.
